Planning to implement eCTD v4.0? Here is what we learned during our Pilot.
-
By DocShifter
- 1 minutes read
A big thank you to Leigh Sandwell, Publishing Regional Director (EU/ROW) at Pfizer, for joining us in our LinkedIn Live session.
With his extensive experience leading global regulatory publishing initiatives, Leigh shared valuable insights from Pfizer’s pilot of eCTD v4.0, and what this next-generation standard means for the future of regulatory submissions.
What did we cover during the session?
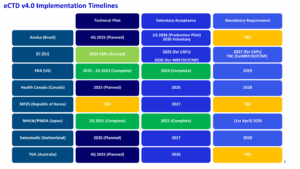
- What are the Health Authority timelines for eCTD v4.0 implementation?
- What are the key benefits and improvements that eCTD v4.0 provides to industry?
- What business impacts have you been considering, and how are you mitigating them?
- How did you prepare for piloting eCTD v4.0 in Pfizer?
- Are there any technical considerations that you needed to address before committing to the pilot?
- What were the outcomes and critical learnings from the pilot?
- How has piloting helped Pfizer prepare for full eCTD v4.0 implementation?
- What are your next steps in becoming implementation-ready?
And many more timely questions were covered during the discussion!
If you missed the session or want to revisit Leigh’s expert guidance, the recording is now available!
Visuals used during the session:
About the guest speaker:
Share this post: